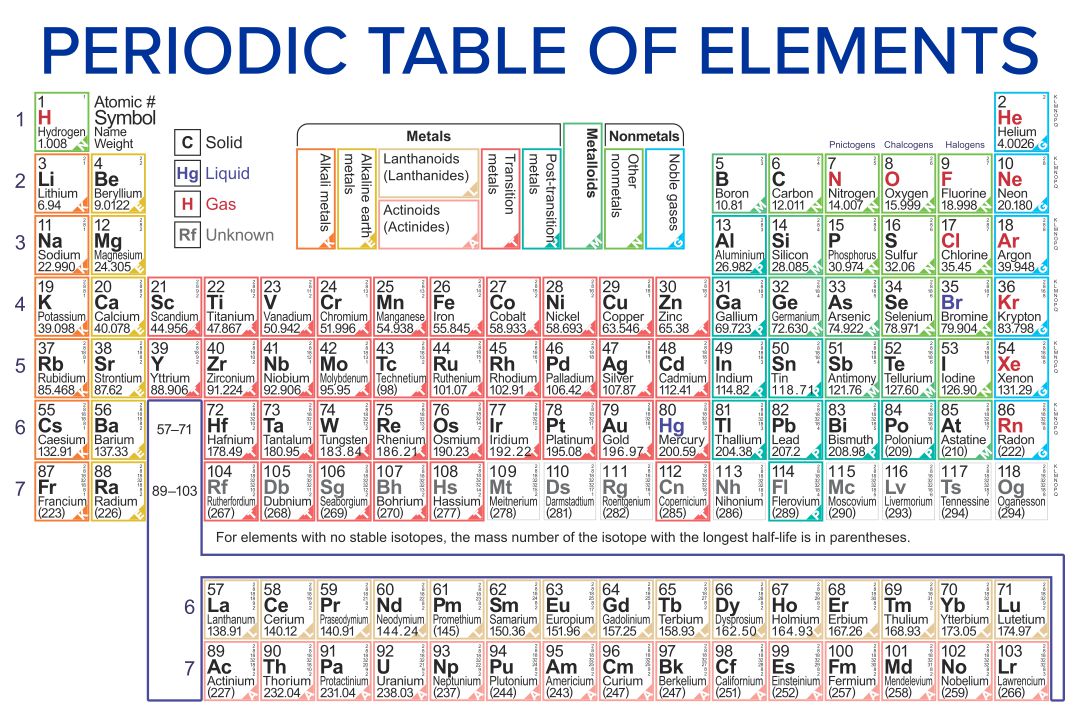
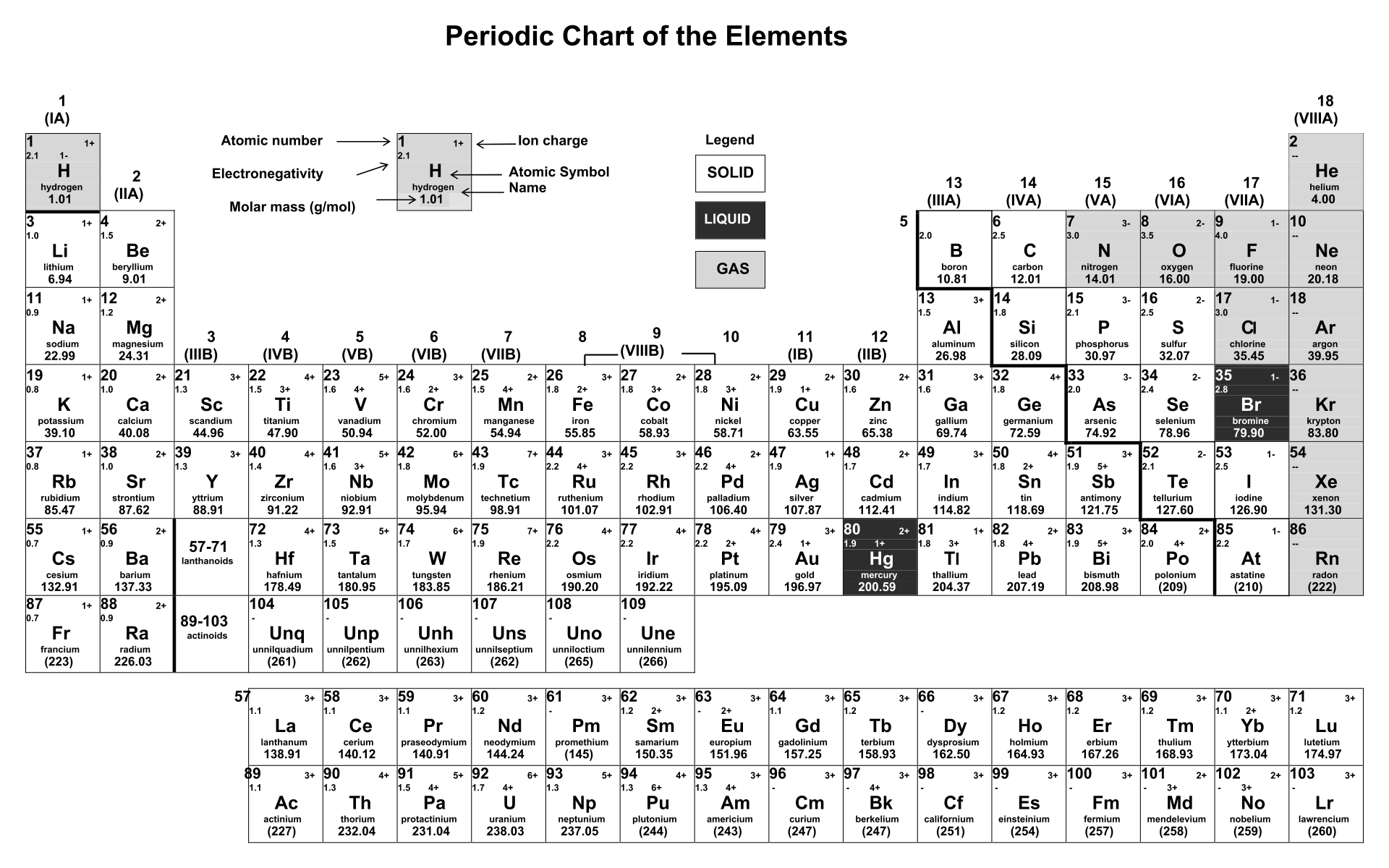
:max_bytes(150000):strip_icc()/PeriodicTableCharge-WBG-56a12db23df78cf772682c37.png)
Periodic Table With Common Ionic Charges
High school chemistry 9 units · 55 skills. Unit 1 Atoms, elements, and the periodic table. Unit 2 Chemical bonding. Unit 3 Chemical reactions. Unit 4 Stoichiometry and the mole. Unit 5 States of matter. Unit 6 Thermochemistry. Unit 7 Solutions, acids, and bases. Unit 8 Reaction rates and equilibrium.

Labeled Periodic Table With Ionic Charges Periodic Table Timeline
Periodic Table with Ions . 3.3 Ionic Bonding. Most of the rocks and minerals that make up the Earth's crust are composed of positive and negative ions held together by ionic bonding. An ionic compound is an electrically neutral compound consisting of positive and negative ions.

Periodic table with charges of ions bezygps
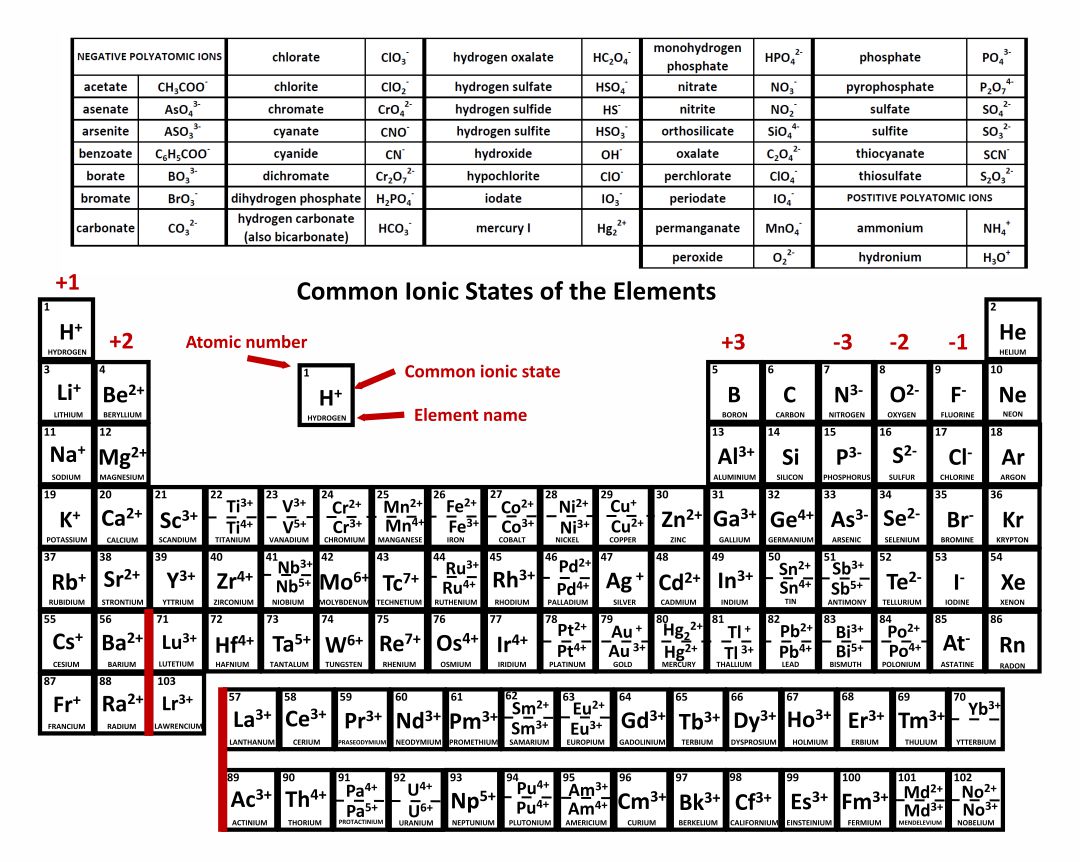
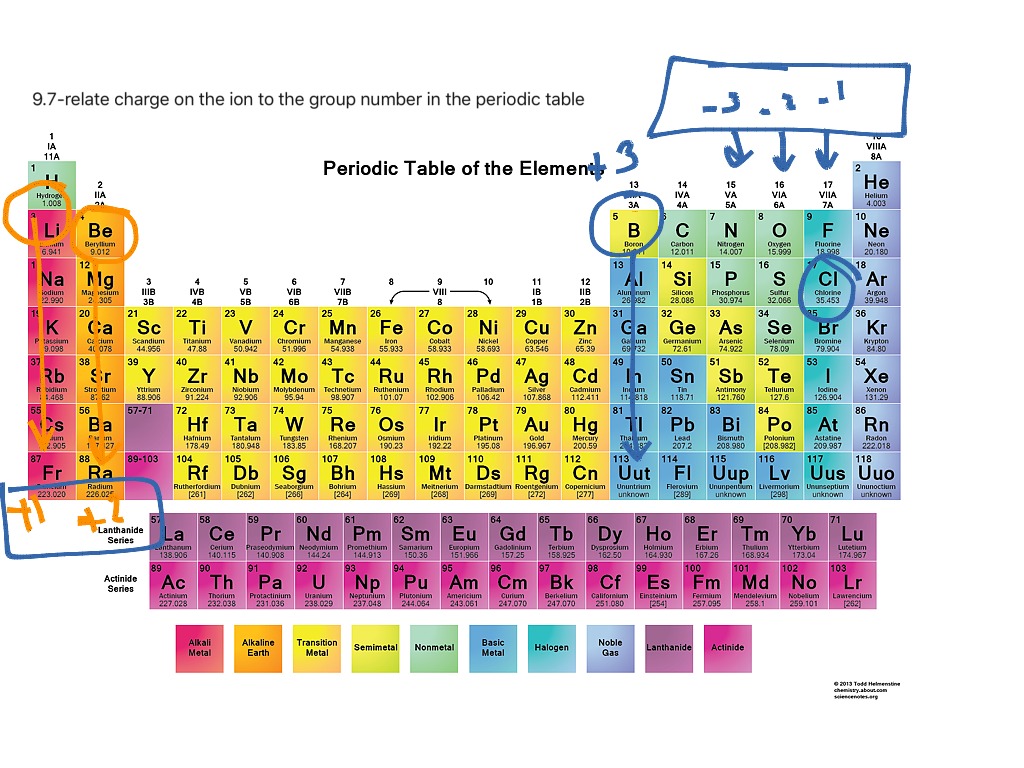
When atoms of nonmetal elements form ions, they generally gain enough electrons to give them the same number of electrons as an atom of the next noble gas in the periodic table. Atoms of group 17 gain one electron and form anions with a 1− charge; atoms of group 16 gain two electrons and form ions with a 2− charge, and so on.

Ions
Atoms are electrically neutral because the number of protons, which carry a 1+ charge, in the nucleus of an atom is equal to the number of electrons, which carry a 1- charge, in the atom. The result is that the total positive charge of the protons cancels out the total negative charge of the electrons so that the net charge of the atom is zero.
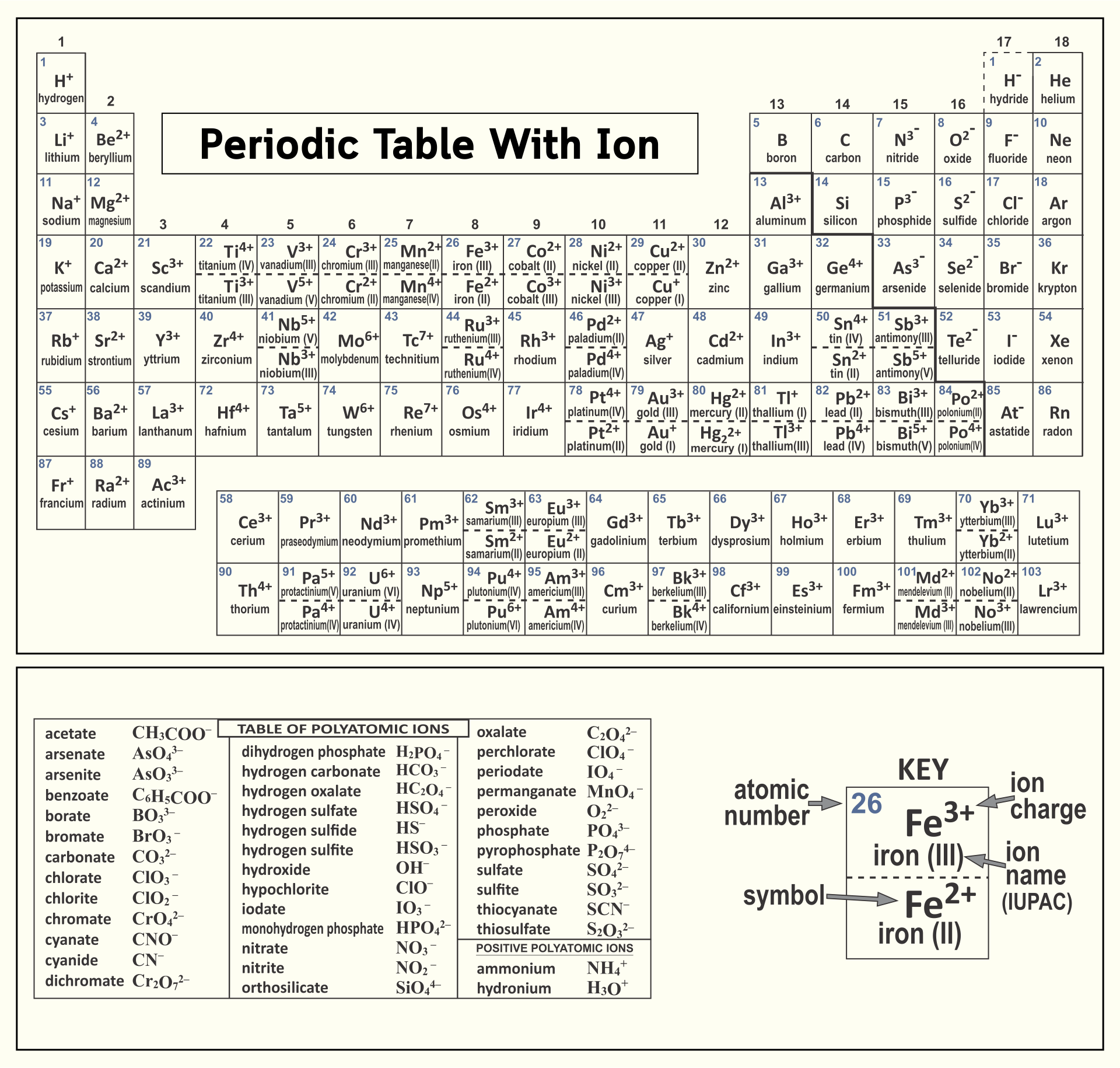
10 Best Printable Periodic Table Of Ions PDF for Free at Printablee
Periodic Table of Elements TABLE LIST W/PROPERTIES GAME Display Property/Trend 17 Cl Chlorine halogen Plot Atomic Mass 1 H Hydrogen nonmetal 2 He Helium noble gas 3 Li Lithium alkali metal 4 Be Beryllium alkaline earth metal 5 B Boron metalloid 6 C Carbon nonmetal 7 N Nitrogen nonmetal 8
Periodic table with charges of ions alertsfas
Learning Objectives By the end of this section, you will be able to: Define ions Identify the charges of the ions formed by main group elements on the periodic table Ions As a recap from Chapter 3, during the formation of some compounds, atoms gain or lose electrons, and form electrically charged particles called ions (Figure 6.1a).
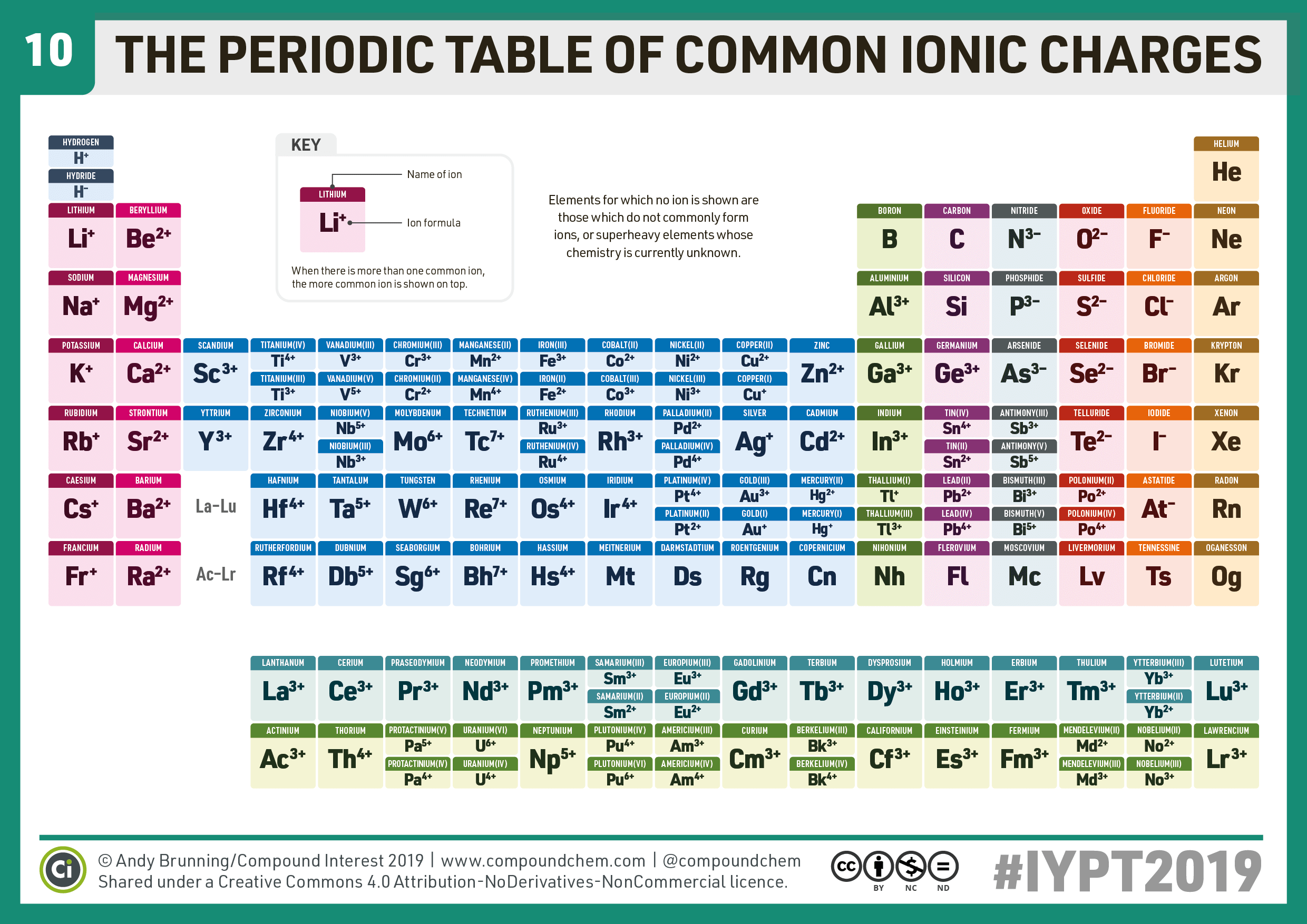
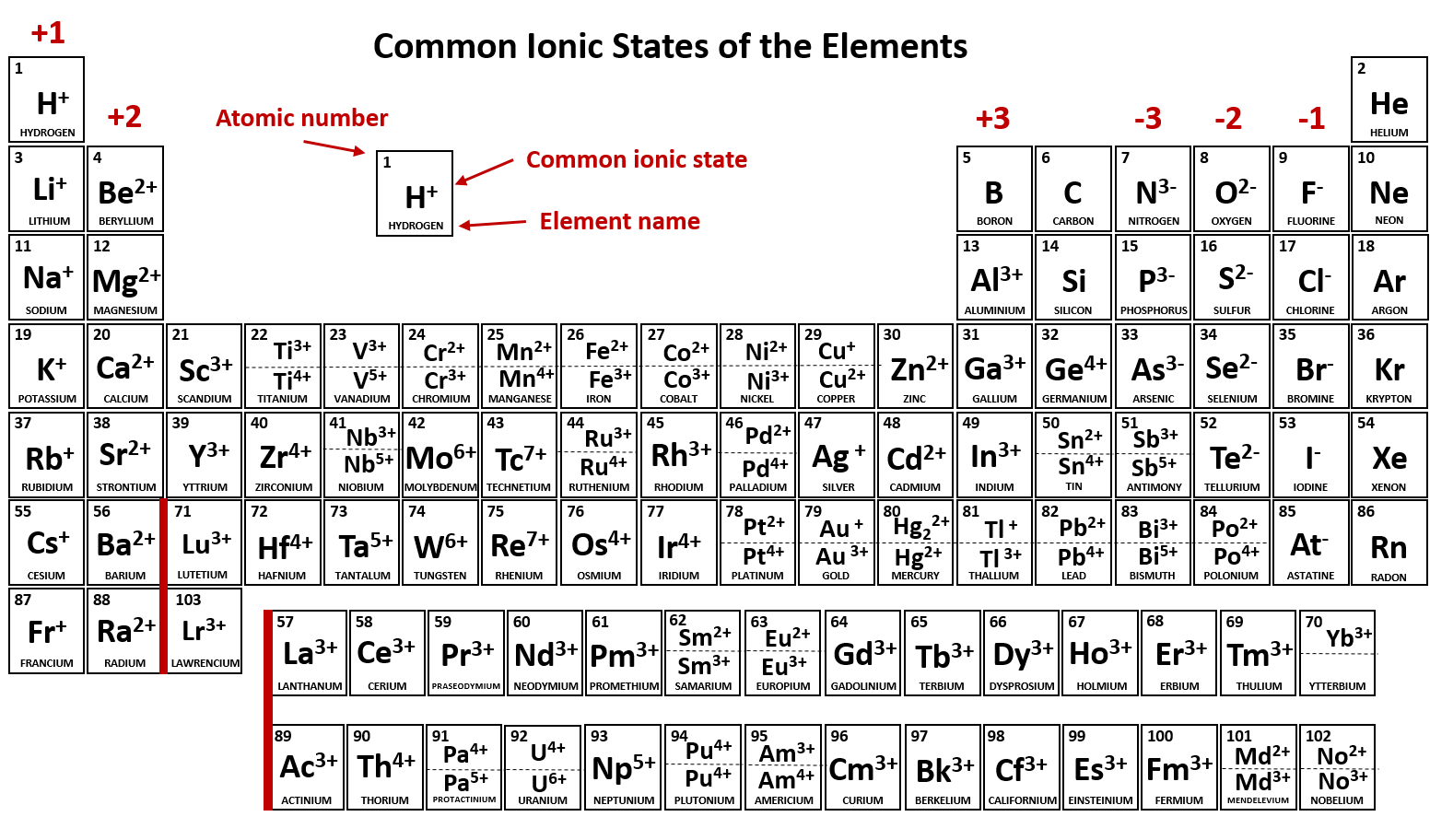
Compound Interest 10 Periodic Table of Common Ions
"An ion is a small electrically charged particle. Ions are single charged atoms (simple ions) or small charged "molecules" (polyatomic ions)." Simple ions include Na +, Ca 2+, and Cl -. Polyatomic ions include (NH 4) +, (CO 3) 2-, and OH -. Unlike protons and neutrons, electrons can be easily removed and added to an atom.
Naming Simple Ionic Compounds Pathways to Chemistry
Compound Interest Explorations of everyday chemical compounds. Home. Infographics Index. Newsletter. About/Contact. Shops. Chem Advent. Click to enlarge. Something a bit more functional for today's periodic table, which looks at the common ions formed by the different elements.
10 Best Printable Periodic Table Of Ions PDF for Free at Printablee
The most-requested printable periodic table lists element charges, to predict compounds and chemical reactions. Now, you can use periodic table trends to predict the most common element charges.

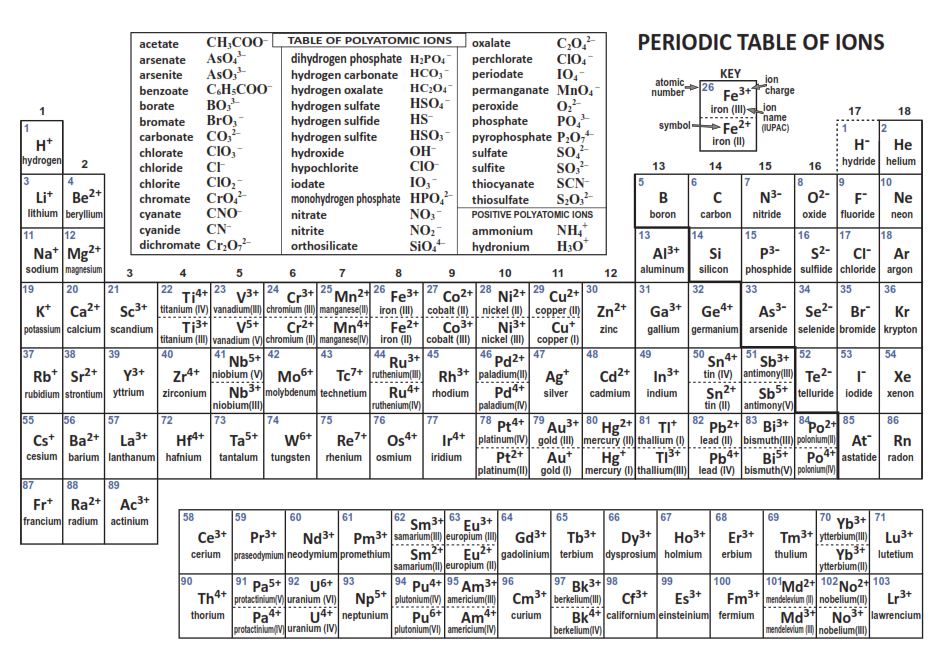
periodic table of ions printable Periodic Chart of Ions PDF Science ) Pinterest
Interactive periodic table showing names, electrons, and oxidation states. Visualize trends, 3D orbitals, isotopes, and mix compounds. Fully descriptive writeups.
Periodic Table Ions List Periodic Table Timeline
Figure 8.2.2 8.2. 2: Definitions of the Atomic Radius. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. (b) The metallic atomic radius, rmet, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as.
periodic table with ions Chemistry
h+. hydrogen. 1. li + lithium. 3. na + sodium. 11. k + potassium. 19. rb + rubidium. 37. cs + cesium. 55. fr + francium. 87. be. 2+ beryllium. 4. mg. 2+ magnesium. 12.
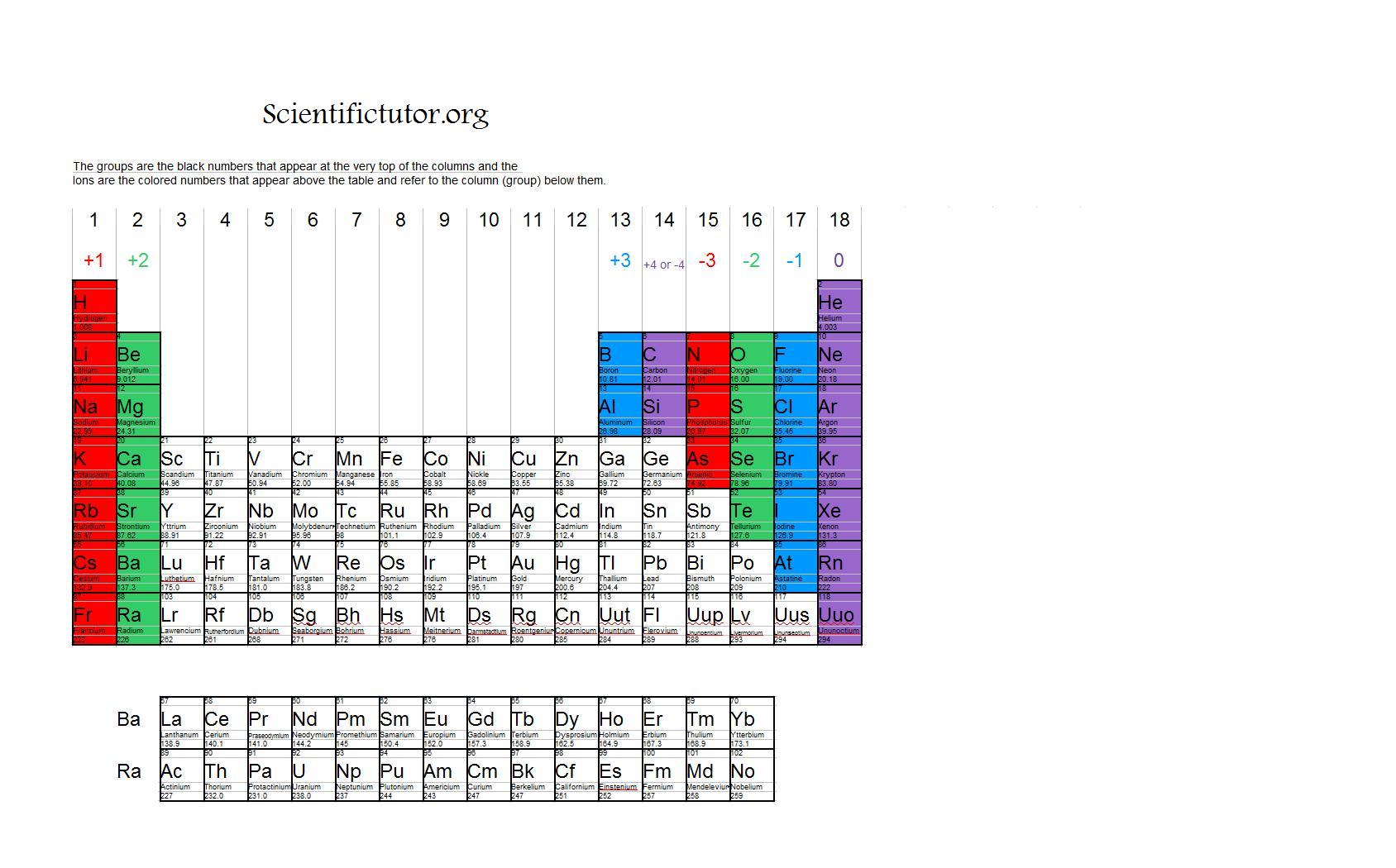
Chem Ions Scientific Tutor
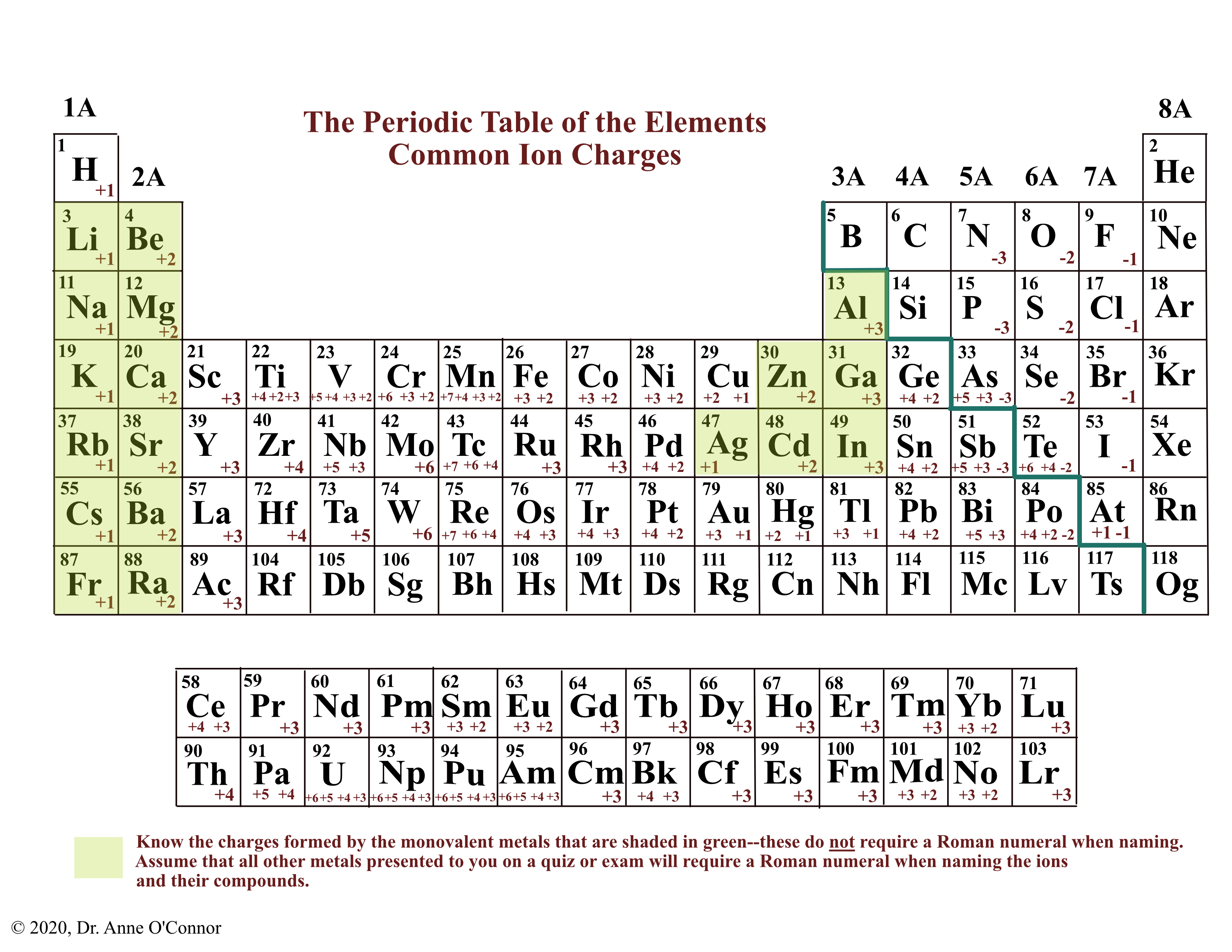
Common charges of ions formed by elements in different groups of the periodic table. Note: Hydrogen is somewhat unusual in that it readily forms both cations and anions. Most elements form only one or the other. Naming cations How do we name main group cations?
10 Best Printable Periodic Table Of Ions PDF for Free at Printablee
Figure \(\PageIndex{3}\): A Plot of Periodic Variation of Atomic Radius with Atomic Number for the First Six Rows of the Periodic Table. In the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column. Because of these two trends, the largest atoms are found in the lower left corner of.
CH150 Chapter 3 Ions and Ionic Compounds Chemistry
Table of Polyatomic Ions. acetate CH 3COO- dichromate Cr 2O 2-. 7 dihydrogen phosphate H 2PO -. 4. ammonium NH4 + cyanide CN- silicate 2- SiO3 benzoate C6H5COO- hydroxide OH- sulphate SO4 2-. borate BO3 3- iodate - IO3 sulphite 2- SO3 carbonate 2- CO3 nitrate - NO3 hydrogen sulphide HS-.

Periodic Table of The Ions PDF Chloride Hydrogen
Note the usefulness of the periodic table in predicting likely ion formation and charge (Figure \(\PageIndex{2}\)). Moving from the far left to the right on the periodic table, main-group elements tend to form cations with a charge equal to the group number. That is, group 1 elements form 1+ ions; group 2 elements form 2+ ions, and so on.